Proton-Coupled Electron Transfer
Project Graphic
Express Summary
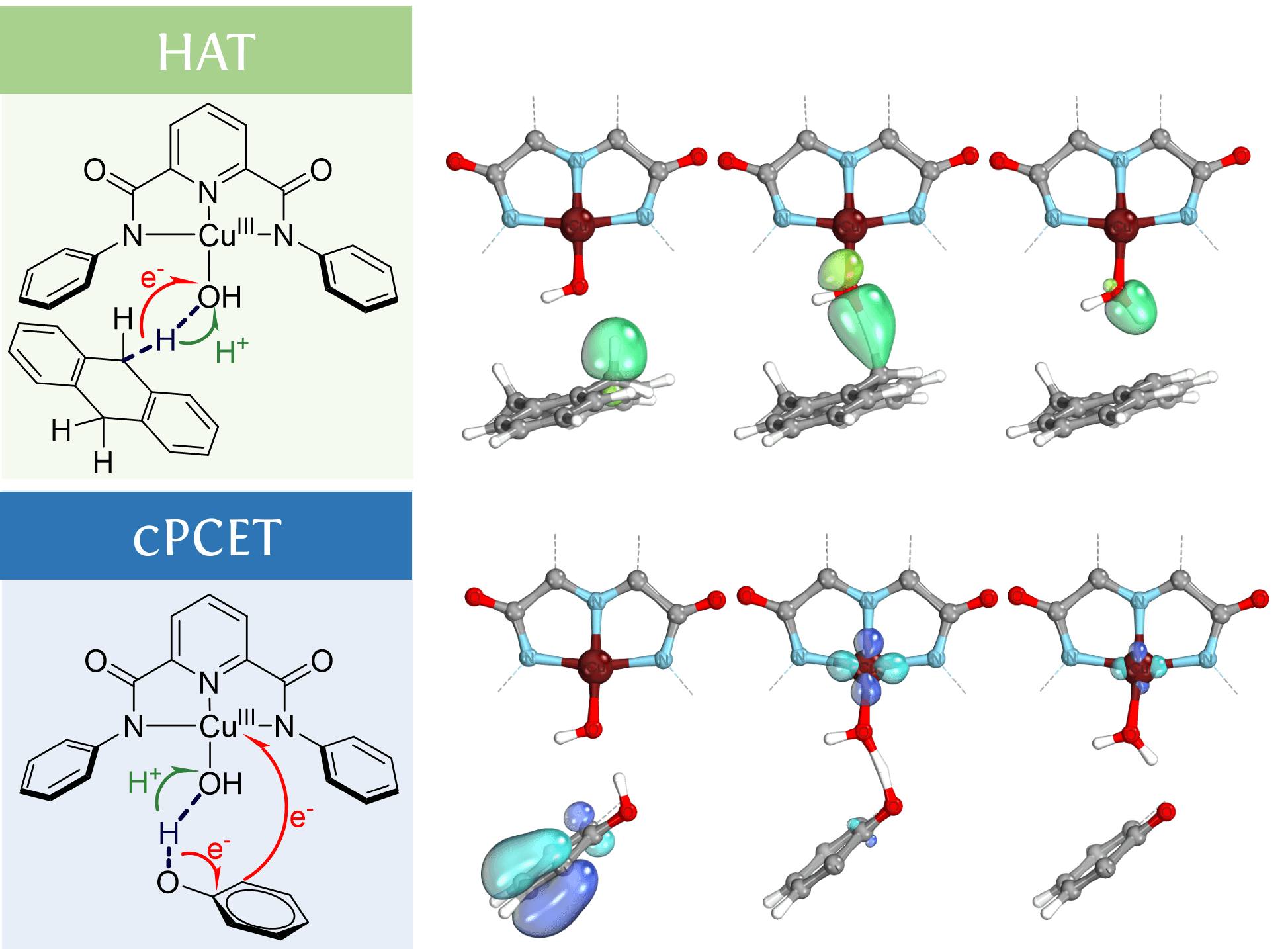
Small-molecule mononuclear copper(III) compounds, such as hydroxo, peroxo, and benzoate, which mimic biological molecules/enzymes, have been found to initiate hydrocarbon oxidation by activating X–H bonds (X = C, O). These reactions can occur through hydrogen-atom transfer (HAT) or concerted proton-coupling electron transfer (cPCET). In our research, we utilize theoretical techniques such as the analysis of the total dipole moment vector oriented along the hydrogen donor-acceptor axis and direct visualization of the localized intrinsic bond orbitals as they evolve along the reaction coordinate to distinguish between HAT and cPCET mechanisms.
Project Summary
Summary of the Project
Project Goals
- Goal-01.
- Goal-02.
- Goal-03.
Relevant Publications
- Pub-01
- Pub-02
- Pub-03
(Social Media Links!)
