Why So Slow? Mechanistic Insights from Studies of a Poor Catalyst for Polymerization of ϵ-Caprolactone
TOC Graphic
Published In
Authors
Daniel E. Stasiw, Mukunda Mandal, Benjamin D. Neisen, Lauren A. Mitchell, Christopher J. Cramer*, and William B. Tolman*
Citation
Stasiw, D. E.; Mandal, M.; Neisen, B. D.; Mitchell, L. A.; Cramer, C. J.; Tolman, W. B. Why so slow? Mechanistic Insights from Studies of a Poor Catalyst for Polymerization of ϵ-Caprolactone. Inorg. Chem. 2017, 56, 725–728. DOI: 10.1021/acs.inorgchem.6b02849
Express Summary
- The polymerization of ε-caprolactone (CL) using an aluminum alkoxide catalyst (Al-TMTAA) was found to have a slower rate compared to other aluminum complexes.
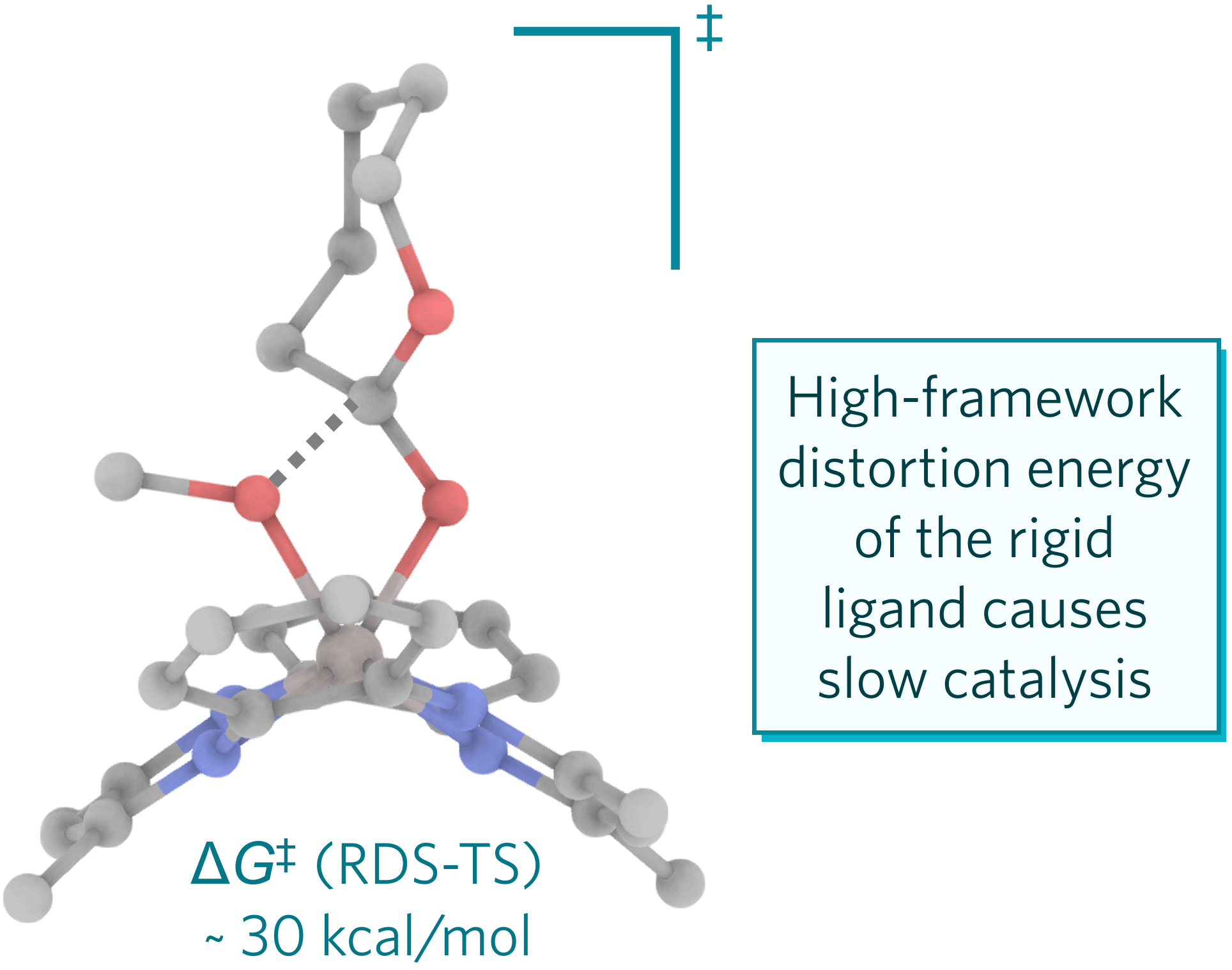
- DFT calculations showed that the ligand framework of the catalyst made trans binding of CL difficult, suggesting a concerted coordination–insertion for polymerization.
- The sluggish performance of the catalyst is attributed to a high-framework distortion energy required to deform the ligand geometry, suggesting a need for ligand flexibility in the design of efficient polymerization catalysts.
Abstract
Polymerization of ε-caprolactone (CL) using an aluminum alkoxide catalyst (1) designed to prevent unproductive trans binding was monitored at 110 °C in toluene-d8 by 1H NMR and the concentration versus time data fit to a first-order rate expression. A comparison of t1/2 for 1 to values for many other aluminum alkyl and alkoxide complexes shows much lower activity of 1 toward polymerization of CL. Density functional theory calculations were used to understand the basis for the slow kinetics. The optimized geometry of the ligand framework of 1 was found indeed to make CL trans binding difficult: no trans-bound intermediate could be identified as a local minimum. Nor were local minima for cis-bound precomplexes found, suggesting a concerted coordination–insertion for polymer initiation and propagation. The sluggish performance of 1 is attributed to a high-framework distortion energy required to deform the “resting” ligand geometry to that providing optimal catalysis in the corresponding transition-state structure geometry, thus suggesting a need to incorporate ligand flexibility in the design of efficient polymerization catalysts.
This Article Contains These Key Ideas
- Idea-01.
- Hypothesis-01.
- Proof-01.
