Mechanism of the Polymerization of rac-Lactide by Fast Zinc Alkoxide Catalysts
TOC Graphic
Published In
Authors
Daniel E. Stasiw, Anna M. Luke, Tomer Rosen, Aaron B. League, Mukunda Mandal, Benjamin D. Neisen, Christopher J. Cramer,* Moshe Kol,* and William B. Tolman*
Citation
Stasiw, D. E.; Luke, A. M.; Rosen, T.; League, A. B.; Mandal, M.; Neisen, B. D.; Cramer, C. J.; Kol, M.; Tolman, W. B. Mechanism of the Polymerization of rac-Lactide by Fast Zinc Alkoxide Catalysts. Inorg. Chem. 2017, 56, 14366–14372. DOI: 10.1021/acs.inorgchem.7b02544.
Express Summary
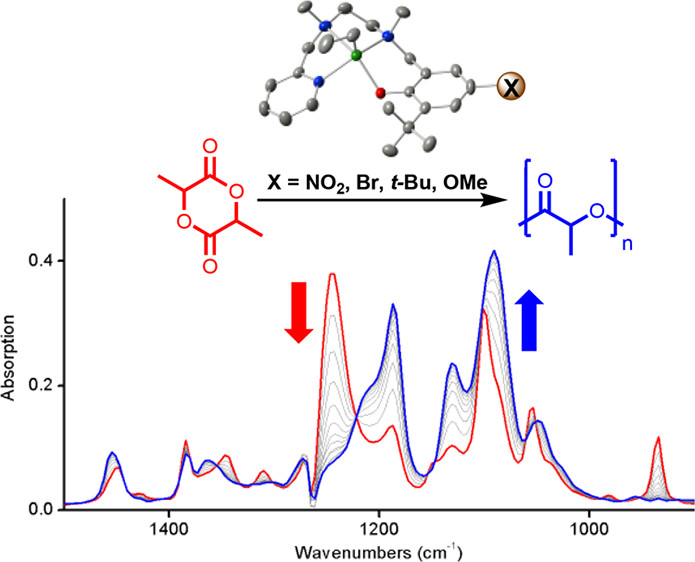
- Polymerization of rac-lactide (rac-LA) using LXZn catalysts was studied using kinetic experiments and DFT calculations.
- LXZn catalysts were effective and relatively selective in polymerizing rac-LA with low dispersities, but the rate of polymerization was less affected by the electronic effects of ligand substituents (controlled by the para substituent X on the bound phenolate donor of the ligand) compared to earlier studies using aluminum catalysts.
- DFT calculations provided insights into the mechanisms of initiation and propagation, helping to explain the high isotacticity and the insensitivity of the rate to the identity of the ligand substituent X.
Abstract
The ring-opening transesterification polymerization (ROTEP) of rac-lactide (rac-LA) using LXZn catalysts (LX = ligand having phenolate, amine, and pyridine donors with variable para substituents X on the bound phenolate donor; X = NO2, Br, t-Bu, OMe) was evaluated through kinetics experiments and density functional theory, with the aim of determining how electronic modulation of the ligand framework influences polymerization rate, selectivity, and control. After determination that zinc-ethyl precatalysts required 24 h of reaction with benzyl alcohol to convert to active alkoxide complexes, the subsequently formed species proved to be active and fairly selective, polymerizing up to 300 equiv of rac-LA in 6–10 min while yielding isotactic (Pm = 0.72–0.78) polylactide (PLA) with low dispersities: Đ = 1.06–1.17. In contrast to previous work with aluminum catalysts for which electronic effects of ligand substituents were significant (Hammett ρ = +1.2–1.4), the LXZn systems exhibited much less of an effect (ρ = +0.3). Density functional calculations revealed details of the initiation and propagation steps, enabling insights into the high isotacticity and the insensitivity of the rate on the identity of X.
This Article Contains These Key Ideas
- Idea-01.
- Hypothesis-01.
- Proof-01.
(Social Media Links!)
